Ph Of 1 M Hcl
pH Calculator
Created by Julia Żuławińska
Reviewed by
Bogna Szyk , Jack Bowater and Adena Benn
Final updated:
Jul 30, 2022
- The pH scale
- Definitions of an acid and a base
- How to discover pH - pH formula
- How to summate pH? - step past step solution
With this pH calculator, y'all tin can determine the pH of a solution in a few means. It can convert pH to H+ , as well as calculate pH from the ionization constant and concentration. The pH value is an essential factor in chemical science, medicine, and daily life. Read the text below to notice out what is the pH scale and the pH formula. In the end, we will also explain how to calculate pH with an easy step-by-stride solution.
Our calculator may ask yous for the concentration of the solution. If you lot don't know, you can calculate it using our concentration computer. You tin also use the solution dilution calculator to calculate the concentration of ions in a diluted solution.
The pH scale
The pH scale (pH) is a numeric calibration used to define how acidic or basic an aqueous solution is. It ordinarily ranges between 0 and 14 but can go beyond these values if sufficiently acidic/basic. The pH value is logarithmically and is inversely related to the concentration of hydrogen ions in a solution. The pH to H+ formula that represents this relation is:
The solution is acidic if its pH is less than 7. If the pH is higher, the solution is basic (also referred to as alkaline). Solutions with a pH that is equal to 7 are neutral.
Apart from the mathematical way of determining pH, yous tin can also use pH indicators. The near universally used pH test is the litmus paper. It changes its color co-ordinate to the pH of a solution in which information technology was dipped. These colors often inspire colorful pH scales:
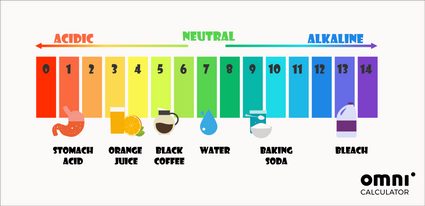
The ph in our bodies is close to neutral. For example, the pH of claret should be around seven.four. The only exception is the stomach, where stomach acids can even reach a pH of 1.
Molecules tin can have a pH at which they are free of a negative accuse. That is what our isoelectric point calculator determines.
Definitions of an acrid and a base
Three unlike theories define acid and base:
- According to the Arrhenius theory, in an aqueous solution, an acid is a substance able to donate hydrogen ions, while a base donates hydroxide ions.
- Brønsted–Lowry theory says that acid can donate protons while a base can accept them.
- Lewis theory states that an acid is something that tin can accept electron pairs. Analogously, a base of operations donates electron pairs.
The college the concentration of hydrogen ions from acid molecules, the lower the pH of the solution and, consequently, the higher its acidity. The reverse is true for hydroxide ions and bases. The higher the concentration of hydroxide ions from base molecules, the higher the pH of the solution and, consequently, the higher its basicity.
Nosotros can describe the reaction of an acid, HA, in water as:
with the acid ionization constant:
A like chemical reaction between base of operations BOH and water looks like this:
The next equation gives the base ionization abiding for the in a higher place formula:
If you lot want to know more about chemic equilibrium constants, check out the equilibrium constant calculator or the reaction quotient calculator.
How to observe pH - pH formula
PH is divers as the negative of the base-ten logarithm of the tooth concentration of hydrogen ions present in the solution. The unit for the concentration of hydrogen ions is moles per liter. To determine pH, you can use this pH to H⁺ formula:
If you already know pH but want to summate the concentration of ions, use this transformed pH equation:
There also exists a pOH scale - which is less popular than the pH scale. pOH is the negative of the logarithm of the hydroxide ion concentration:
or
pH and pOH are related to one another by this pOH and pH equation:
How to calculate pH? - step past step solution
- Let'due south assume that the concentration of hydrogen ions is equal to 0.0001 mol/L.
- Calculate pH by using the pH to H⁺ formula:
- Now, you tin can also easily determine pOH and a concentration of hydroxide ions using the formulas:
Of form, you lot don't have to perform all of these calculations by mitt! Choose the option to decide pH with ion concentration in the calculator, and blazon in any of these four values! Then, picket as the tool does all the work for you!
- Alternatively, yous tin can discover a chemical from the lists (of acids or bases). Permit'southward say you want to know how to find the pH of formic acrid -
HCOOH. ItsKais0.00018. - Choose the concentration of the chemical. Let's assume that information technology'southward equal to
0.1 mol/L. - To find a concentration of H⁺ ions, you have to...:
where
Here
-
cis the tooth concentration of the solution; and -
xis equal to the molar concentration of H⁺.
For 0.1 Thou HCOOH:
Now you know how to calculate pH using pH equations. If yous notice these calculations time-consuming, feel gratis to utilise our pH calculator. Select your chemical and its concentration, and watch it practise all the work for you. When y'all're finished, check out the titration calculator!
I want calculate the pH from..
Ph Of 1 M Hcl,
Source: https://www.omnicalculator.com/chemistry/ph
Posted by: adamsrequithe.blogspot.com


0 Response to "Ph Of 1 M Hcl"
Post a Comment